Vul deze aan.
Leerdoelen
•Je kunt de namen van moleculaire stoffen opstellen.
•Je kunt de molecuulmassa van moleculen berekenen.
•Je kunt het massapercentage van atomen in een molecuul berekenen.
Moleculaire stoffen en zouten: naamgeving
In de scheikunde gebruiken we systematische namen om stoffen een duidelijke naam te geven. Dat is de scheikundige naam. Deze namen vertellen je direct welke atomen erin zitten en hoeveel van elk.
Systeemnaam: telwoorden en uitgangen
Voor moleculaire verbindingen gebruiken we telwoorden om aan te geven hoeveel atomen van een bepaalde soort in het molecuul aanwezig zijn. Deze telwoorden komen uit het Grieks. Als het eerste element in de naam maar één keer voorkomt, gebruiken we daarvoor geen 'mono'.
Getal | Telwoord |
|---|---|
1 | mono |
2 | di |
3 | tri |
4 | tetra |
5 | penta |
Het laatste element in de naam krijgt meestal een speciale uitgangsnaam, vaak eindigend op '-ide'. Denk aan zuurstof (O), dat als laatste element 'oxide' wordt, of chloor (Cl), dat 'chloride' wordt.
Voorbeelden:
•CO2: koolstofdioxide (di = 2 zuurstofatomen)
•P2O5: difosforpentoxide (di = 2 fosforatomen, penta = 5 zuurstofatomen)
Stof | Uitgangsnaam |
|---|---|
O | oxide |
Cl | chloride |
S | sulfide |
F | fluoride |
I | jodide |
P | fosfide |
Verschil met zouten
Zouten, ook wel ionaire verbindingen genoemd, krijgen geen telwoorden. Dat komt omdat je bij zouten met verhoudingen werkt. Door te kijken naar de positieve en negatieve ladingen van de ionen (plusjes en minnetjes), weet je precies hoeveel van elk ion er nodig is om een neutrale verbinding te vormen. Bij moleculaire stoffen kan dat niet zomaar, en daarom zijn daar de telwoorden zo belangrijk.
De massa van een molecuul
Elk atoom heeft een eigen massa, afhankelijk van het aantal protonen en neutronen. Een molecuul, dat uit meerdere atomen bestaat, heeft daardoor ook een massa. We noemen dit de molecuulmassa. De grootheid hiervan is M (hoofdletter M) en de eenheid is u (atomaire massa-eenheid). Eén u is 1,7\cdot10^{-27}1,710^{-27}1,7x10^{-27}1,7x10^{-27}71,7x10^{-27}271,7x10^{-27}^{-}271,7x10^{-2}^{-}271,7x10^{-}^{-}271,7x10^{}^{-}271,7x10^1^{-}271,7x10^{10}^{-}271,7x10^{-}271,7x10^{^{\placeholder{}}-}271,7x10^{-^{\placeholder{}}-}271,7x10^{\placeholder{}^{\placeholder{}}-}27 kg.
Molecuulmassa berekenen
De massa van ieder atoom kun je opzoeken in je Binas. Linksboven in elk vakje van een element in het periodiek systeem staat de gemiddelde atoommassa in u.
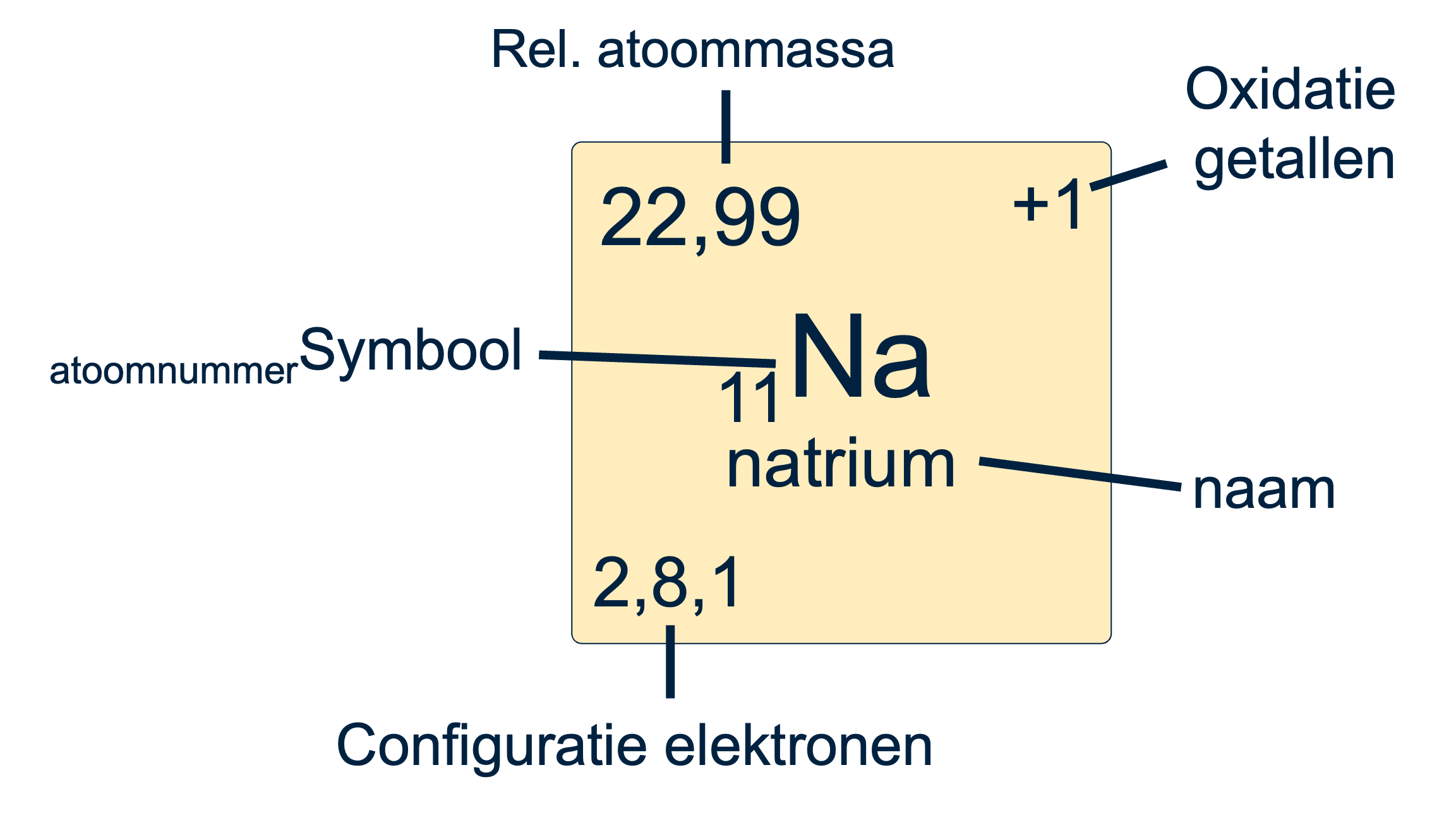
Om de molecuulmassa te berekenen, tel je de massa's van alle atomen in het molecuul bij elkaar op. Voor zouten moet je eerst zelf de juiste verhoudingsformule maken voordat je kunt berekenen hoeveel van elk atoom erin zit.
Rekenvoorbeeld: Molecuulmassa van P2O5 (difosforpentoxide)
1.Zoek de atoommassa's op in Binas:
P2O5 | ||
|---|---|---|
Element | Index | M |
Fosfor (P) | 2 | 31 u |
Zuurstof (O) | 5 | 16 u |
2. Tel de massa's bij elkaar op:
2 x P-atomen: 2 * 31 u = 62 u |
5 x O-atomen: 5 * 16 u = 80 u |
Totale molecuulmassa van P2O5 = 62 u + 80 u = 142 u |
Massapercentage van atomen
Soms wil je weten welk deel van de totale massa van een molecuul uit een specifiek atoom bestaat. Dit noemen we het massapercentage. Je berekent dan hoeveel procent van de totale massa van een molecuul afkomstig is van een bepaald atoom.
Stappenplan massapercentage berekenen
1.Bereken de totale massa van het hele molecuul (de molecuulmassa) in u.
2.Bereken de totale massa van het specifieke element dat je wilt onderzoeken in u.
3.Gebruik de volgende formule om het massapercentage te berekenen: Massapercentage = (massa van het element in u / totale molecuulmassa in u) x 100%
\text{Massapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{aMassapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{aMssapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{assapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%M\text{assapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{assapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{massapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{massapercentage = }\frac{\left(\text{massa element in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\text{massapercentage = }\frac{\left(\text{massa element in u}\right)}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\&\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\&\%\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\&\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\&\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot100\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot10\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot1\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\cdot\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in }}\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in}}\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{i}}\text{massapercentage = }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage =}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage -}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage - }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage -}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage }\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentage}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentag}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercenta}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercent}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercentr}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercent}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massapercen}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massaperce}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massaperc}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massaper}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massape}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massap}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{massa}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{mass}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{mas}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{ma}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\text{m}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}m\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}ma\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}mas\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}mass\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massa\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massap\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massape\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massaper\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massaperc\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massaperce\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercen\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuu}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{i}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{i}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in }}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercenta\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercentag\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercentage\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercentage=\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercentage\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercentag\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercenta\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercent\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massapercen\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massaperce\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massaperc\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massaper\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massape\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massap\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}massa\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}mass\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}mas\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}ma\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}m\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}M\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}Ma\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}Mas\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}Ma\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}M\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in u}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in }}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{in}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}\text{i}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}i}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}in}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}in}u\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}in}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}i}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuuli}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuulin}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuuli}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}n}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}ni}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}n}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}^{^{\placeholder{}}}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuu}^{^{\placeholder{}}}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}^{^{\placeholder{}}}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}^{\placeholder{}^{\placeholder{}}}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}^{\placeholder{}}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul }}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuul}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecuu}}}\frac{\text{massa element in u}}{\text{M}_{\text{molecu}}}\frac{\text{massa element in u}}{\text{M}_{\text{molec}}}\frac{\text{massa element in u}}{\text{M}_{\text{mole}}}\frac{\text{massa element in u}}{\text{M}_{\text{mol}}}\frac{\text{massa element in u}}{\text{M}_{\text{mo}}}\frac{\text{massa element in u}}{\text{M}_{\text{m}}}\frac{\text{massa element in u}}{\text{M}_{\placeholder{}}}\frac{\text{massa element in u}}{\text{M}}\frac{\text{massa element in u}}{\placeholder{}}\text{massa element in u}\text{massa element in }\text{massa element in}\text{massa element i}\text{massa element }\text{massa element}\text{massa elemen}\text{massa eleme}\text{massa elem}\text{massa ele}\text{massa elel}\text{massa elelm}\text{massa elelme}\text{massa elelmen}\text{massa elelment}\text{massa elelment }\text{massa elelment}\text{massa elelmen}\text{massa elelme}\text{massa elelm}\text{massa elel}\text{massa ele}\text{massa el}\text{massa e}\text{massa }\text{massa}\text{mass}\text{mas}\text{ma}\text{m}m\text{M}mmamamasmassmass\text{ }elmass\text{ }elemmass\text{ }elememass\text{ }elemenmass\text{ }elementmass\text{ }elementdmass\text{ }element\frac{mass\text{ }element}{\placeholder{}}mass\text{ }elementmass\text{ }\frac{element}{\placeholder{}}mass\text{ }elementmass\text{ }element5mass\text{ }elementmass\text{ }element5mass\text{ }elementmass\text{ }element\in\frac{mass\text{ }element\in}{}\frac{mass\text{ }element\in}{\placeholder{}}\frac{massa\text{ }elementi}{\placeholder{}}\frac{massa\text{ }element}{\placeholder{}}\frac{massaelement}{\placeholder{}}\frac{massaelemen}{\placeholder{}}\frac{massaeleme}{\placeholder{}}\frac{massaelem}{\placeholder{}}\frac{massaele}{\placeholder{}}\frac{massael}{\placeholder{}}\frac{massae}{\placeholder{}}\frac{massa}{\placeholder{}}\frac{massa}{\placeholder{}}e\frac{massa}{\placeholder{}}el\frac{massa}{\placeholder{}}ele\frac{massa}{\placeholder{}}el\frac{massa}{\placeholder{}}e\frac{massa}{\placeholder{}}\frac{mass}{\placeholder{}}\frac{mas}{\placeholder{}}\frac{ma}{\placeholder{}}\frac{m}{\placeholder{}}\frac{\placeholder{}}{\placeholder{}}5
Deze formule is eigenlijk hetzelfde als "deel gedeeld door geheel keer honderd procent", die je misschien kent van economie.
Rekenvoorbeeld: massapercentage chloor (Cl) in aluminiumchloride (AlCl3)
Aluminiumchloride (AlCl3) is een zout, dus we moeten eerst de verhoudingsformule bepalen.
Verhoudingsformule opstellen:
1.Aluminium (Al) heeft als ion Al3+.
2.Chloor (Cl) heeft als ion Cl-.
3.Om een neutrale verbinding te krijgen (evenveel plusjes als minnetjes), hebben we één Al3+ ion en drie Cl- ionen nodig.
4.De verhoudingsformule is dus AlCl3.
Het massapercentage van AlCl3 berekenen:
Stap 1: Bereken de molecuulmassa van het hele molecuul (AlCl3).
1.Zoek de atoommassa's op in Binas:
atoommassa's | |
|---|---|
Aluminium (Al) | 27 u |
Chloor (Cl) | 35,5 u |
2. Tel de massa's bij elkaar op om de molecuulmassa van AlCl3 te berekenen:
Molecuulmassa AlCl3 |
|---|
1 x Al = 27 u |
3 x Cl = 3 * 35,5 u = 106,5 u |
Totaal = 27 u + 106,5 u = 133,5 u |
Stap 2: Bereken de totale massa van het element chloor in AlCl3.
3 x Cl = 3 * 35,5 u = 106,5 u |
Stap 3: Gebruik de formule voor het massapercentage.
\text{Massapercentage chloor = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage chloor = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage chloor = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage chloo = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage chlo = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage chl = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage ch = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage c = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mAlCl}_{\text{3}}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mAlCl}_{\placeholder{}}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mAlCl}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mAlC}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mAl}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mA}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{m}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A\text{m}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{m}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A;}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A;}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{A}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{m}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mo}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mol}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{mole}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{molec}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{molecu}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{molecuu}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{3 }}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa ACl}_{\text{3 }}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa AlCl}_{\text{3 }}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlCl}_{\text{3 }}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlCl}_{\text{3}}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlCl}_{\placeholder{}}\text{in u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlClin u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlCin u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAlin u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massaAin u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massain u}\right)}{\left(\text{M}_{\text{molecuul}}\text{in u}\right)}\cdot100\%
\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\thickapprox80\%\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\thickapprox80\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\thickapprox8\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\thickapprox\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%=\text{Massapercentage chloor }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage chloo }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage chlo }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage chl }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage ch }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage c }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage C }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot100\%\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot100\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot10\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot1\text{Massapercentage }=\frac{106{,}5}{133{,}5}\cdot\text{Massapercentage }=\frac{106{,}5}{133{,}5}\text{Massapercentage }=\frac{106{,}5}{133{,}}\text{Massapercentage }=\frac{106{,}5}{133}\text{Massapercentage }=\frac{106{,}5}{13}\text{Massapercentage }=\frac{106{,}5}{1}\text{Massapercentage }=\frac{106{,}5}{\placeholder{}}\text{Massapercentage }=106{,}5\text{Massapercentage }=106{,}\text{Massapercentage }=106\text{Massapercentage }=106.\text{Massapercentage }=106.5\text{Massapercentage }=106.\text{Massapercentage }=106\text{Massapercentage }=10\text{Massapercentage }=105\text{Massapercentage }=10\text{Massapercentage }=1\text{Massapercentage }=\text{Massapercentage }=\text{ }\text{Massapercentage }=\text{ }\text{Massapercentage }=\text{ 2}\text{Massapercentage }=\text{ }\text{Massapercentage }=\text{ }\text{Massapercentage }=\text{Massapercentage}=\text{Massapercentage}\text{Massapercentage }\text{Massapercentage =}\text{Massapercentage }\text{Massapercentage}\text{Massapercentage }\text{Massapercentage }\text{Massapercentage =}\text{Massapercentage }\text{Massapercentage }\text{Massapercentage}\text{Massapercentage }\text{Massapercentage}\text{Massapercentag}\text{Massapercenta}\text{Massapercent}\text{Massapercen}\text{Massaperce}\text{Massaperc}\text{Massaper}\text{Massapere}\text{Massaperex}\text{Massaperex}\text{Massaper}\text{Massape}\text{Massap}\text{Massa}\text{Mass}\text{Mas}\text{Ma}\text{M}MMaMaMasMassMassMassaMassapeMassaperMassaperMassapercMassapercMassapercentaMassapercentaMassapercentagMassapercentageMassapercentage=MassapercentageMassapercentagMassapercentagMassapercentaMassapercentMassapercenMassaperceMassapercMassaperMassapeMassapMassaMassMasMaM\text{Ma}\text{Mas}\text{Mas}\text{Mass}\text{Massap}\text{Massape}\text{Massaper}\text{Massaperc}\text{Massaperce}\text{Massaperce}\text{Massapercent}\text{Massapercenta}\text{Massapercenta}\text{Massapercentage}\text{Massapercentage }\text{Massapercentage =}\text{Massapercentage = }\text{Massapercentage = 1}\text{Massapercentage = 10}\text{Massapercentage = 1}\text{Massapercentage = }\frac{\text{Massapercentage = }}{\placeholder{}}\text{Massapercentage = }\text{Massapercentage = }5\text{Massapercentage = }\frac{\text{Massapercentage = }}{\placeholder{}}\text{Massapercentage = }\text{Massapercentage = }\left(\right.\text{Massapercentage = }\left(\text{m}\right.\text{Massapercentage = }\left(\text{ma}\right.\text{Massapercentage = }\left(\text{ma}\right.\text{Massapercentage = }\left(\text{mass}\right.\text{Massapercentage = }\left(\text{massa}\right.\text{Massapercentage = }\left(\text{massa}\right.\text{Massapercentage = }\left(\text{massa}\right.\text{Massapercentage = }\left(\text{massa }\right.\text{Massapercentage = }\left(\text{massa C}\right.\text{Massapercentage = }\left(\text{massa Cl}\right.\text{Massapercentage = }\left(\text{massa Cl}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\text{i}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\text{in}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\text{in }\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\text{in u}\right.\text{Massapercentage = }\left(\text{massa Cl}_{\text{ }}\text{in u}\right)\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{}\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\cdot\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\cdot1\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\cdot10\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\cdot100\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{A}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{Al}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{Al}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{Al}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{i}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in}\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in }\right.}\cdot100\%\text{Massapercentage = }\frac{\left(\text{massa Cl}_{\text{ }}\text{in u}\right)}{\left(\text{M}_{\text{AlCl}_{\text{3}}}\text{in u}\right.}\cdot100\%
Dus, ongeveer 80% van de massa van aluminiumchloride bestaat uit chloor.